What You Should Know About CSV in Pharma
Por um escritor misterioso
Descrição
Learn more about computer system validation, which is required by the FDA and other global regulatory bodies for drug and medical device manufacturers.

Computer System Validation (CSV) in Life Sciences Part 1: Introduction to CSV - Verista

SOP for Computer System Validation in Pharmaceutical Industry

Importance of computer system validation (CSV) in Pharmaceutical Industry

Complete guide to computer system validation in 2023

Basics of Computerized System Validation in Pharma Industry, CSV
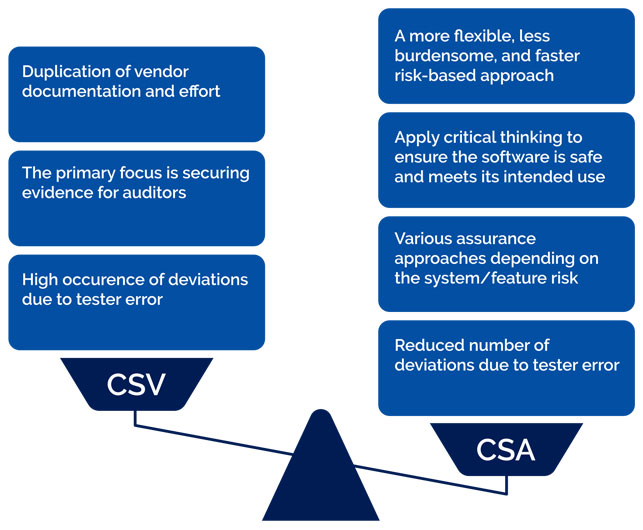
What is Computer Software Assurance (CSA), the FDA's new approach to CSV? - Vimachem

LIMS Validation Plan

Computer System Validation in Pharmaceutical Industry

COMPUTER SYSTEM VALIDATION [CSV] - pharma arena

The Complete Guide to Computer System Validation: IQ, OQ, PQ, – Blue Mountain

Computer System Validation in Pharmaceutical Industry - Veeprho

Data Integrity in the Pharmaceutical Industry

PDF) Computer system validation in the perspective of the pharmaceutical industry
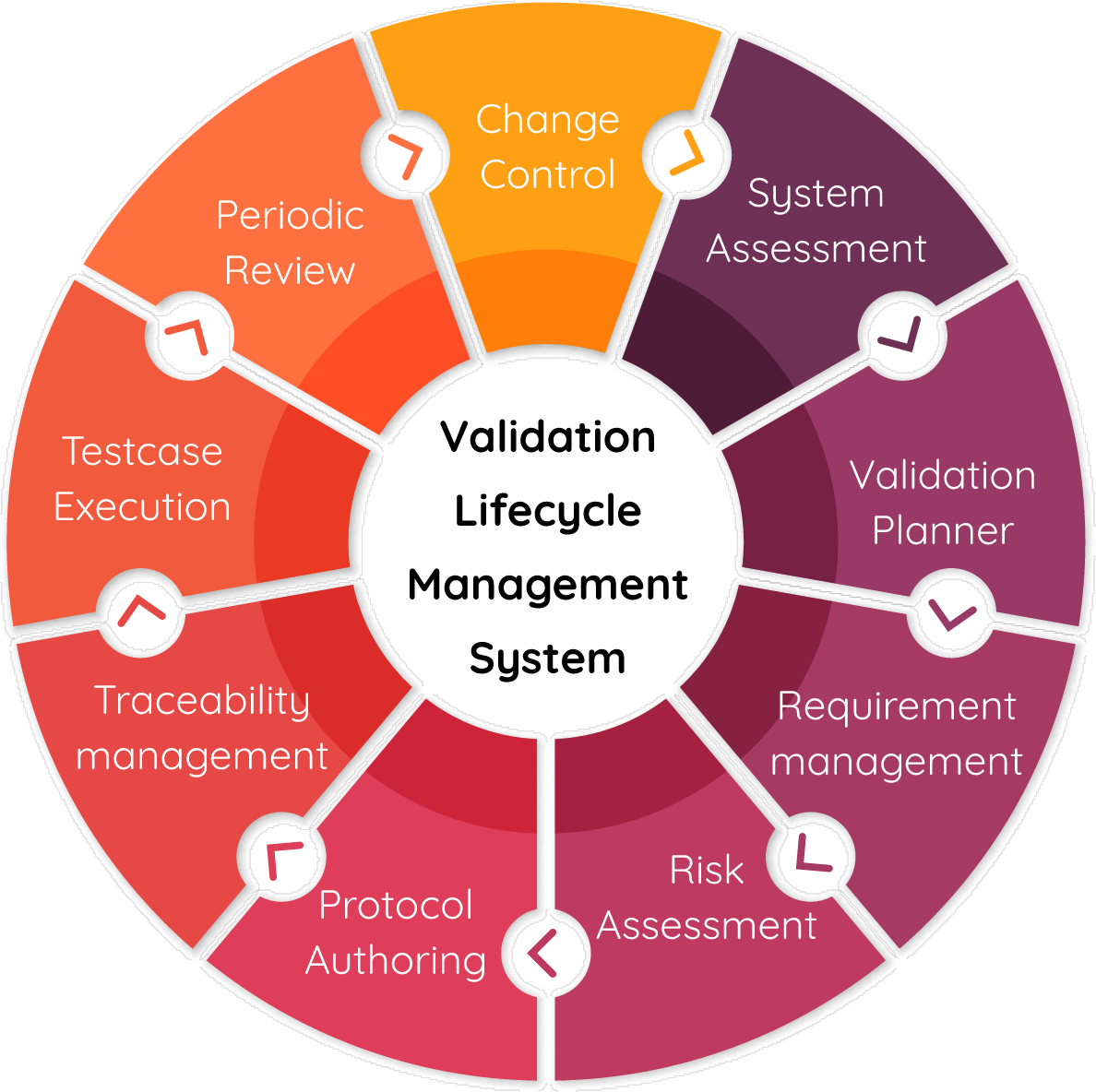
Computer System Validation (CSV) Services, Clinical Services, Services
de
por adulto (o preço varia de acordo com o tamanho do grupo)




:max_bytes(150000):strip_icc()/avengersendgame5cbe860b2f312-1-2000-7d9142bd8bab44eb87628b0c08ce0ea6.jpg)
